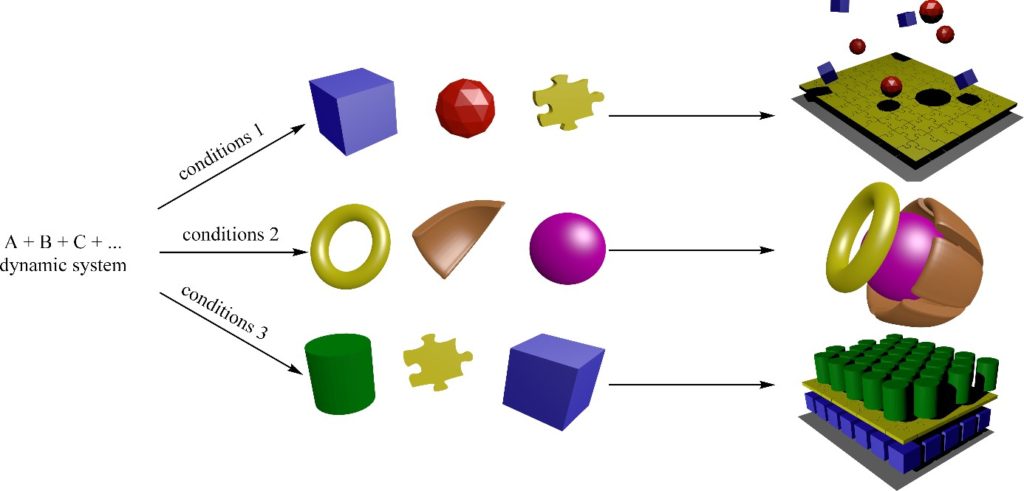
Dynamic reaction networks
Synthesis of individual components of a library that upon mixing forms all possible combinations. All reactions, however, are coupled by sharing their reactants/product thus forming a reaction network. Each particular product features distinct properties that can be addressed by external stimuli, making it adaptive. And adaptation is the key prerequisite for evolution.
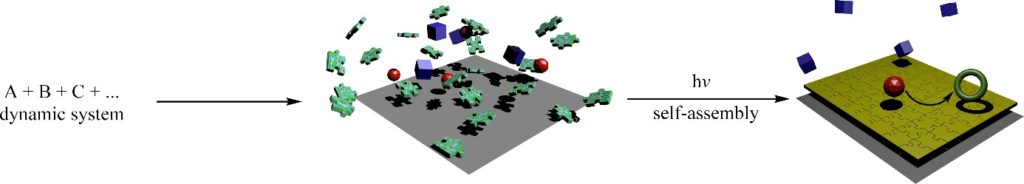
Self-assembly at surfaces and interfaces

Introducing a phase interface, e.g. a solid surface, inherently imposes a bias on the system. Due to differences in surface adsorption enthalpies among the species present, the composition of the layer adsorbed on the surface will be complementary to that in the other phase. A phase interface represents a significant energy step for physisorbing molecules comparable to the activation energy of a reversible bond formation/exchange barrier. We aim to understand how the surface instruct self-organization of species in its proximity and how molecules ‘feel’ the presence of the surface.
Heterogeneous photo-organo-catalysis
Molecules and the surface that they are assembled on exchange energy and electrons so that excited states can travel from the substrate to the molecule (or vice versa). Molecular excited state can adopt many roles such as a reagent or a catalyst. Organic molecules in their vast structural variations can recognize each other and therefore can be potent organocatalysts. We want to ‘shed the light’ on organocatalysis at surfaces… literally!
Surfaces for biochemical and biomedical applications

Every cell is defined by its boundary with the world around – cell membrane – which is an amazingly complex ‘surface’, an example of a triple liquid phase interface. We are interested in surfaces which interact with biomolecules, cellular structures or even whole cells and can be used as sensors of these biomolecules or for cellular biology studies of the structures (e.g. membranes). By studying the phase boundary we break down inter-disciplinary boundaries!